When you are picking up a medication – either a tablet, capsule, or liquid medication – you likely focus on the condition it treats and not how it got to you. However, there is a detailed process behind every pharmaceutical product consisting of three main components: Active Pharmaceutical Ingredient (API), Finished Dosage Form (FDF), and formulation manufacturing.
Even though understanding the terms may feel overwhelming, you do not need to have a scientific background to know what they mean. In this article, we will explain these terms in a straightforward way so that you will understand how a medicine is developed and how it is eventually sold, as well as the role that Lifevision Healthcare plays in this process.
What is an Active Pharmaceutical Ingredient?
The Active Pharmaceutical Ingredient, or API, is the main part of the medicine. The API is the material that produces the desired therapeutic effect when prescribed or taken by the patient.
To understand this better, consider an example:
If a tablet were considered a meal, then the API would be the main course of the meal, such as rice or bread; it is what provides the basic function.
Examples: For instance, the API for a tablet that relieves pain is likely to be the ingredient used (Paracetamol); for a tablet used to treat an infection, the API will be the substance used to kill the bacteria that cause the patients’ infection.
Some reasons why caring about the API is important:
- It gives an indication of how well the medication or drug product will be able to perform its intended function(s).
- It defines the intended purpose of the drug.
- It must have specific qualities and characteristics, and must always meet the quality and safety specifications outlined in the relevant regulatory framework (i.e., the respective country and/or region in which the drug is going to be distributed).
In the absence of an API, there will not be a finished drug product.
What are finished dosage forms (FDFs)?
After the active pharmaceutical ingredient (API) has been manufactured, it is typically not used directly to treat patients; instead, the API must undergo additional processing in order to create a suitable medication referred to as an FDF.
In layman’s terms, an FDF is the drug product purchased at your local pharmacy.
FDF examples include:
- Injections
- Creams or ointments
What are the reasons that you would not use an API directly?
The API generally has the following characteristics when produced:
- Too potent in its raw state
- Difficult to accurately measure
- Not stable or easy to take
For these reasons, other ingredients are added to the API during the manufacturing process to create a safe and effective drug product that is easy to take.

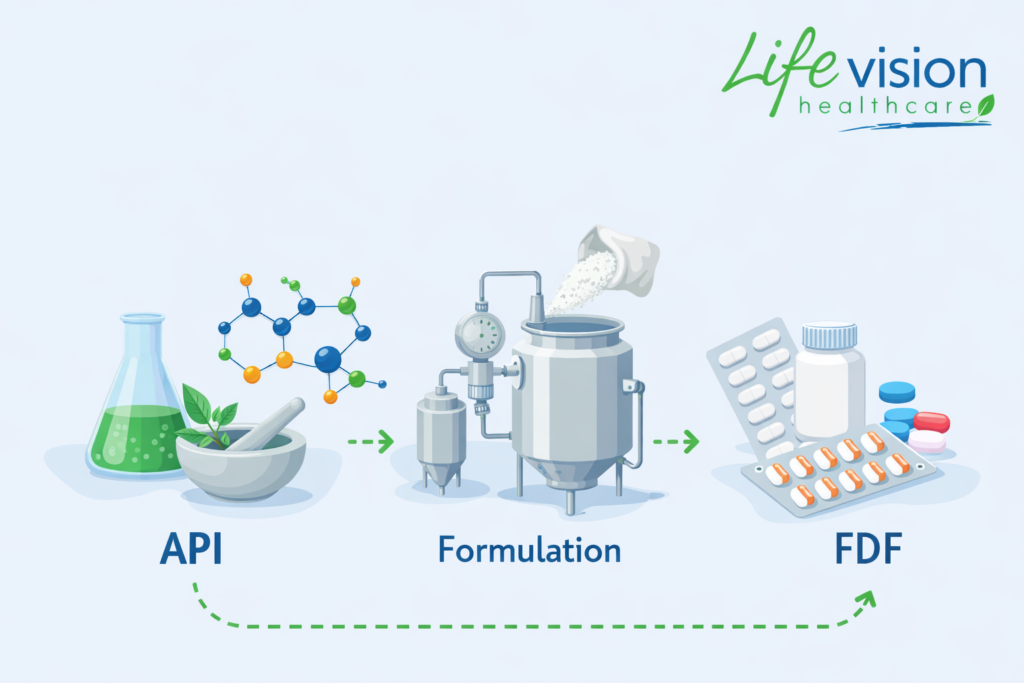
What is formulation manufacturing?
Formulation manufacturing is the process of transforming the API into a finished dosage form (FDF). This is where science and form meet.
During the formulation process, the following things happen:
The API is combined with other ingredients (excipients)
Excipients are inert ingredients that help with the following:
- Stability of the drug
- Taste masking of the drug
- Controlling the release of the drug within the body
The dosage form is designed based on how the dosage form meets the needs of the patient (i.e. tablet, syrup, or injection).
The final product is tested for safety and efficacy.
Final product meets all regulatory requirements and the efficacy is verified through testing.
The final product will be packaged and distributed. By properly packaging and distributing the drug, it is ensured that the drug is delivered safely to the end user.
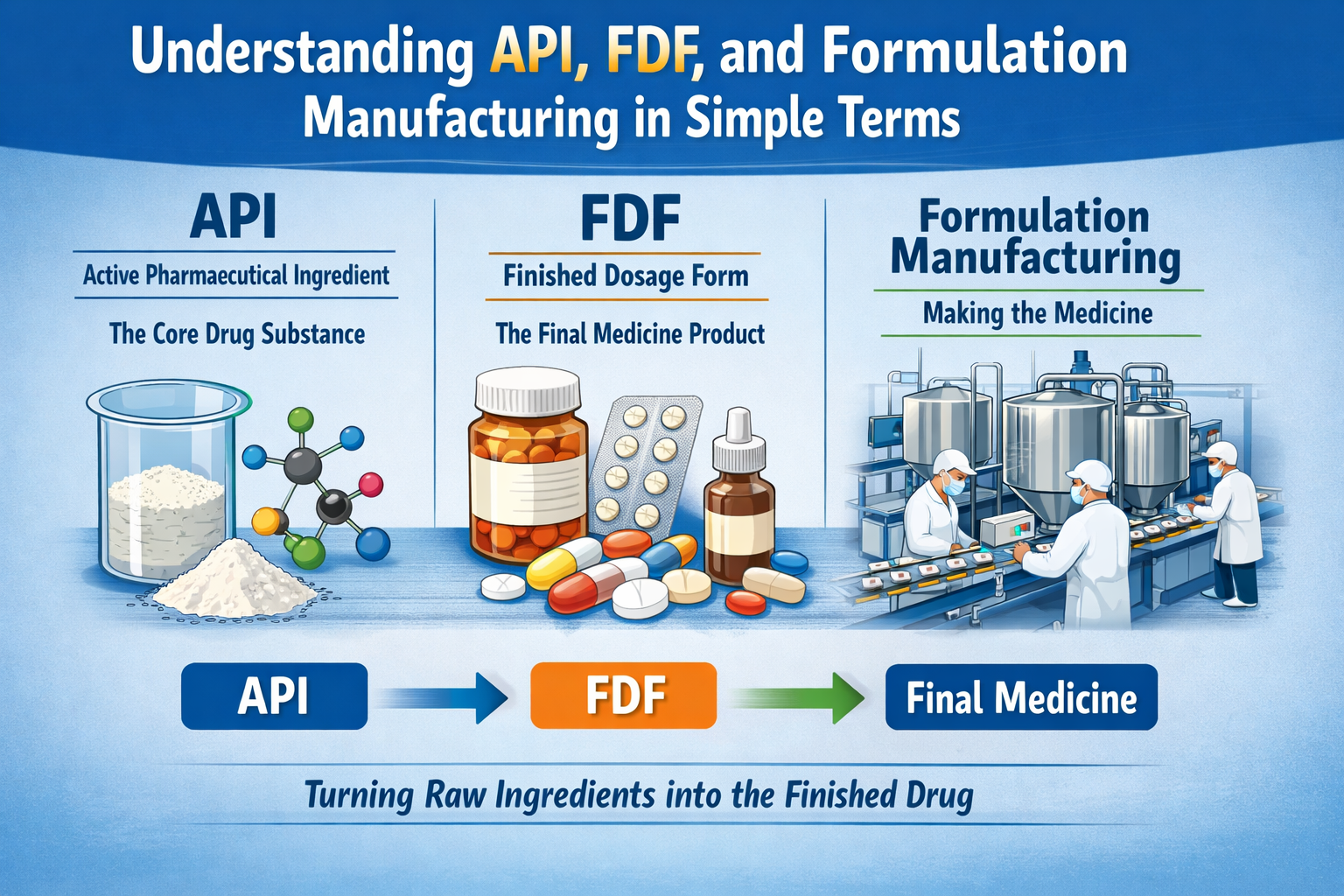
The Connection Between API & FDF And Their Relationship to Formulation
The API, FDF, and formulation process are all interconnected as follows:
- The API is the substance in a drug that creates the therapeutic benefit.
- The formulation process is what takes the raw API and makes it into a product that can be used.
- The FDF is the drug that will actually be taken by the patient.
To use an analogy:
API = engine
Formulation = assembly of a car
FDF = finished car ready to be driven.
If any one of these three is missing, then drugs cannot be effectively used by the patient.
Why This Is Important
Although the process may seem simple, understanding it can help explain why pharmaceutical manufacturing is so highly regulated and closely monitored.
Reasons include:
- Patient Safety: Poorly formulated products can be potentially dangerous or ineffective as drugs.
- Consistency: Each dose of the drug should have the same effect regardless if you receive one today or 3 months from now.
- Efficiency: A properly formulated product will work as intended in your body.
- Compliance: Pharmaceutical manufacturers must adhere to strict regulations in order to maintain global standards.
Lifevision Healthcare’s Role In Pharmaceutical Manufacturing
Lifevision Healthcare is a company involved in successfully manufacturing and marketing high-quality pharmaceutical products at every stage of this process.
We are focused on:
- Creating reliable, valid, and safe formulations
- Ensuring continuous quality throughout the entire manufacturing process
- Providing diverse product lines for our customers
- Partnering with customers to provide consistent and trusted medicines.
Lifevision Healthcare’s product line illustrates our technical capability to formulate and manufacture drugs.
We are a provider of high-quality pharmaceuticals and manages the manufacturing processes as part of our overall commitment to delivering superior products.
What Lifevision Healthcare Offers:
- Safe & effective formulations.
- Quality assurance is maintained throughout the entire process, from development through distribution and delivery.
- Lifevision Healthcare provides a complete range of products that meet patient needs.
- A range of products demonstrates the company’s capabilities in the areas of formulation and manufacturing.
- A wide range of products covers a variety of therapeutic categories, thereby allowing distributors and providers to better serve our patients.
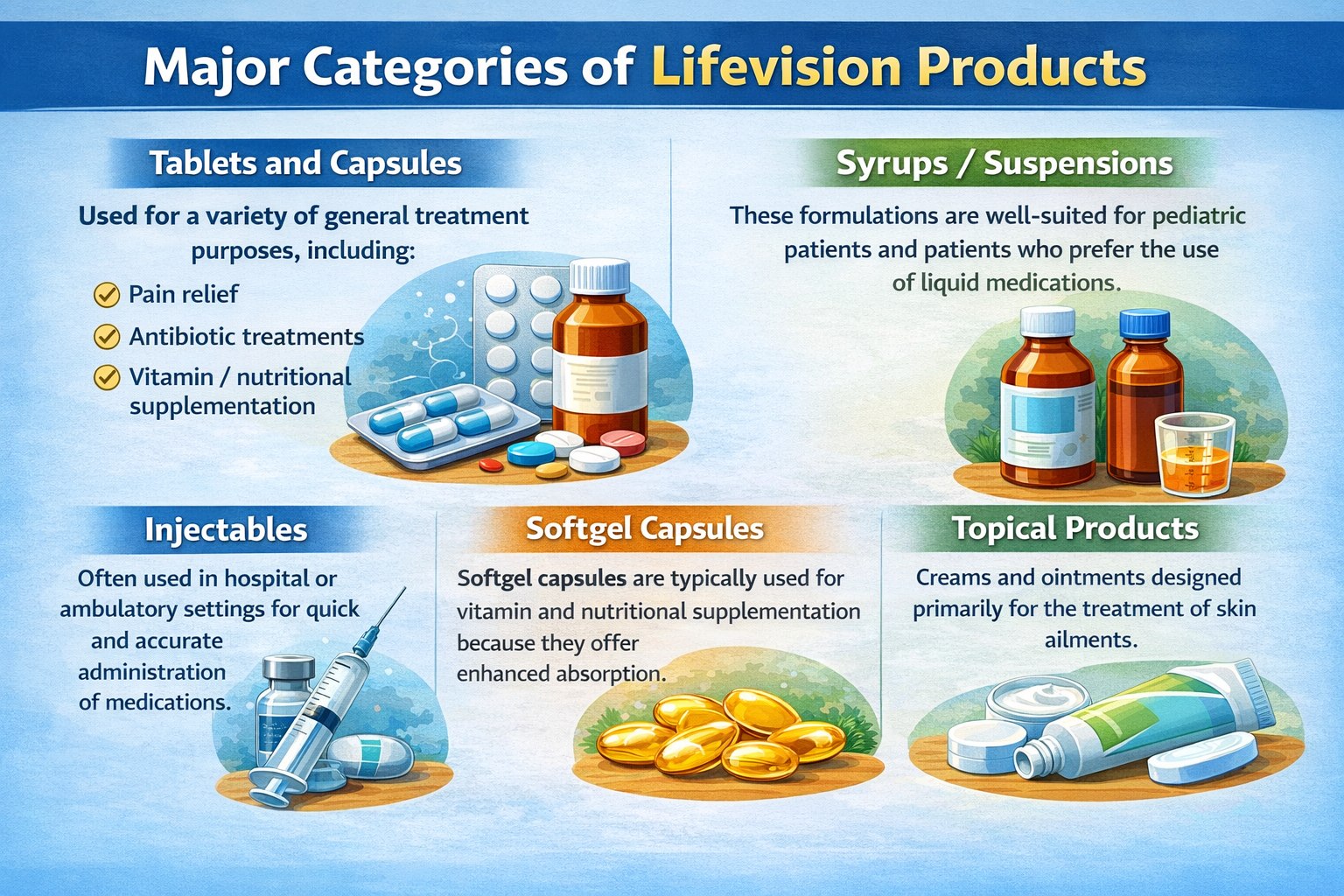
Major Categories of Lifevision Products
- Tablets and Capsules
Used for a variety of general treatment purposes, including:
- Pain relief
- Antibiotic treatments
- Vitamin / nutritional supplementation
- Syrups / Suspensions
These formulations are well-suited for pediatric patients and patients who prefer the use of liquid medications.
- Injectables
Often used in hospital or ambulatory settings for quick and accurate administration of medications.
- Softgel Capsules
Softgel capsules are typically used for vitamin and nutritional supplementation because we offer enhanced absorption.
- Topical Products
Creams and ointments designed primarily for the treatment of skin ailments.
How API and Formulation Affect Product Quality
The quality of a medicine depends heavily on both API and formulation.
Key factors:
- Purity of API
- Stability of the formulation
- Accuracy of dosage
- Manufacturing standards
Companies like Lifevision Healthcare play a crucial role in this ecosystem by delivering a wide range of high-quality products that meet modern healthcare needs. Our diverse product portfolio also creates excellent opportunities for internal linking, helping businesses improve both usability and search engine performance.
The manufacturing process for pharmaceuticals continues to be adapted and improved through the emergence of trends that change how APIs and FDFs are manufactured. Some of these trends include: advanced drug delivery systems; personalised medicine; improved methods for increasing bioavailability; and sustainable and environmentally friendly manufacturing practices. Manufacturers continuously seek to innovate and find improved methods to provide better patient care and to meet the demand for medicines in healthcare.
Summary!
In short, while understanding the manufacturing process of APIs, FDFs, and formulations can seem cumbersome, all you need to know at the end of the day is this: the API is what makes the medicine work; the FDF is what you use to take the medicine, and when you combine both, the result is an FDF ready for consumption. Together, the API, FDF and formulation represent the foundation of the pharmaceutical manufacturing process.
FAQs for API, FDF, and Formulation Manufacturing in Simple Terms:
What’s the difference between an active Pharmaceutical Ingredient (API) and a Finished Dosage Form (FDF)?
The active pharmaceutical ingredient (API) is the component within pharmaceuticals that provides their therapeutic effects; the finished dosage form (FDF) would thus represent the medicinal products that are finally provided to a patient, such as tablets or syrups.
Why can’t patients take active pharmaceutical ingredients (APIs) directly?
This is due to the fact that, as “active” pharmaceutical ingredients, APIs are typically either too strong in raw form (potent) or too volatile (unstable), by themselves, to be safely delivered to patients in their appropriate dosages without being mixed with other ingredients to ensure the integrity and suitability of those dosages for use by patients.
What does manufacturing formulation entail?
The manufacturing formulation process is finding the appropriate combination of active pharmaceutical ingredients (APIs) with excipients, determining the appropriate dosage form for those active pharmaceutical ingredients (APIs), testing the safety of the mixture, and producing the final product for distribution to patients.
How does the formulation of a drug affect its efficacy?
The primary influence formulation will have on the efficacy of a drug is through the decisions made upon how the drug will be absorbed into a patient’s bloodstream, as well as how, after being absorbed, it will be released into a patient’s system, and then subsequently utilised by a patient’s body.
What types of products does Lifevision Healthcare develop?
Lifevision Healthcare develops an extensive variety (tablets, capsules, syrups, injectables, softgels, and topical) of finished product pharmaceuticals that represent the therapeutic requirements of many therapeutic segments and various healthcare needs.






 Call Us: 8062750200
Call Us: 8062750200 Contact Us
Contact Us